Understanding Market Exclusivity and Why It Matters for Your Launch

Why One Per Market Exclusivity Determines Who Wins After Launch
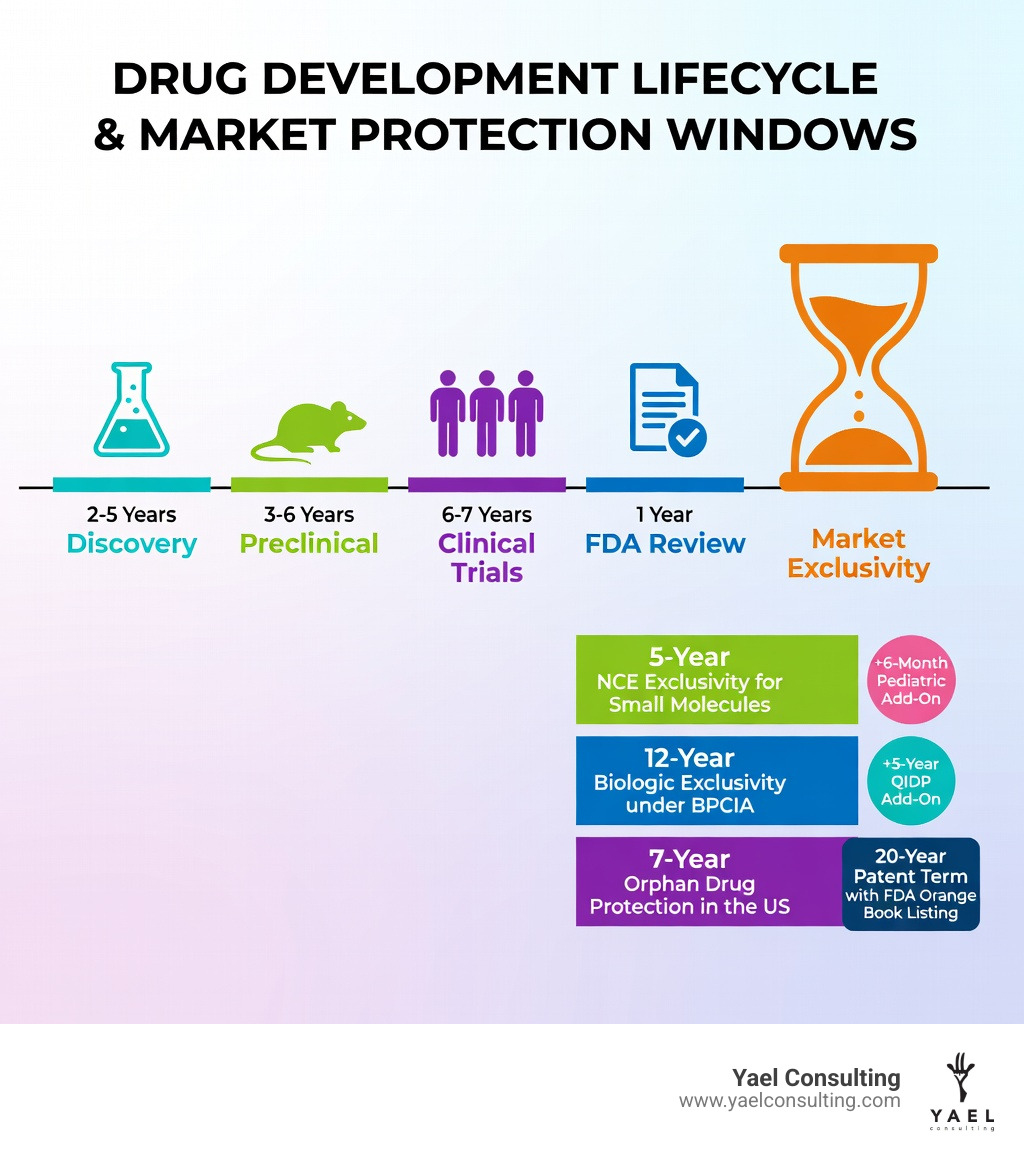
One per market exclusivity is a period of legal protection that prevents competitors from selling the same drug or biologic after it receives regulatory approval. Here’s a quick breakdown:
| Type | Duration | Who It Protects |
|---|---|---|
| New Chemical Entity (NCE) | 5 years | First-in-class small molecules |
| Biologic Exclusivity | 12 years | Biologic reference products |
| Orphan Drug | 7 years (US) / 10 years (EU) | Rare disease treatments |
| Clinical Investigation | 3 years | New uses or delivery methods |
| Pediatric Extension | +6 months | Drugs with pediatric studies |
| QIDP (Antibiotic) | +5 years | Qualifying infectious disease drugs |
This protection is separate from patents. It’s granted by the FDA upon approval — and it can make or break a drug’s commercial success.
Most newly approved drugs enjoy more than 12 years free from generic competition. First-in-class drugs average around 14.5 years. That window is when companies recover R&D costs, build market share, and generate the revenue that funds future innovation.
But understanding which exclusivity applies to your drug — and how to maximize it — is where most companies leave money on the table.
I’m Lior Krolewicz, a marketing strategist who has helped businesses across industries protect and leverage their competitive positioning — including applying the principle of one per market exclusivity to agency relationships, guaranteeing each client they’re the only business in their market we represent. That same strategic thinking shapes how I help clients plan launches that dominate before competitors can respond.
What is Market Exclusivity and How Does it Differ from Patents?
In high-stakes product launches, we often hear people use “patents” and “exclusivity” interchangeably. However, they are two very different beasts. Think of it like this: a patent is your deed to a house (property rights), while market exclusivity is the zoning law that says no one else can build a similar house on your block for a set amount of time (marketing prohibitions).
Statutory Rights: USPTO vs. FDA
Patents are property rights granted by the U.S. Patent and Trademark Office (USPTO). They can be issued at any point during a drug’s development and usually last 20 years from the date of filing. They cover the “how” and “what” of your invention—the chemical structure, the way it’s manufactured, or how it’s used.
Exclusivity, on the other hand, is a statutory right granted by the FDA under the Federal Food, Drug, and Cosmetic Act (FD&C Act). It only attaches upon approval of a drug. While patents can be challenged in court or bypassed through clever engineering, exclusivity is a hard stop. It prevents the FDA from even accepting or approving a competitor’s application for a specific period. For more details, you can read How to understand Patents and Exclusivity.
Concurrent Protection
The magic happens when these two work together. They can run concurrently, or exclusivity can extend your protection after a patent has expired. In some cases, a drug might not even have a strong patent, but it can still enjoy a robust monopoly because of FDA-granted one per market exclusivity.
| Feature | Patent | Regulatory Exclusivity |
|---|---|---|
| Granted By | USPTO | FDA |
| Timing | Any time during development | Only upon drug approval |
| Duration | 20 years from filing | 180 days to 12 years |
| Focus | Intellectual Property | Marketing Rights |
| Legal Basis | Patent Law | FD&C Act / BPCIA |
The Role of the Orange Book in Market Protection
If you are launching a drug in the US, your “Bible” is the “Approved Drug Products with Therapeutic Equivalence Evaluations,” commonly known as the Orange Book.
Listing Requirements and Generic Notification
The FDA is required by law to list every patent number and expiration date for approved drugs in the Orange Book. When a generic company wants to launch a version of your drug, they have to look at the Official Orange Book Database and make a “certification” regarding your patents.
If they challenge your patent (a “Paragraph IV certification”), it can trigger a 30-month stay of approval, giving you time to defend your market position. Interestingly, the FDA doesn’t send you a gold-embossed letter when you get exclusivity; they just update the Orange Book. It’s the official vehicle for letting the world know that your market is closed to outsiders.
Navigating the US Framework: Types of One Per Market Exclusivity
The US framework is designed to balance two competing needs: the need to reward innovators for the massive costs of R&D (often $1-2 billion per drug) and the need to eventually allow low-cost generics into the market. This balance was famously struck by the Hatch-Waxman Act of 1984.
To understand how to protect your launch, we need to look at the specific types of 21 CFR 314.108 Regulations that govern small molecules and biologics.
New Chemical Entity (NCE) and Clinical Investigation Exclusivity
The “Gold Standard” for small molecule protection is the 5-year NCE exclusivity. This is granted to a drug that contains an active moiety that has never been approved by the FDA before.
- 5-Year NCE: During this time, the FDA cannot even accept an Abbreviated New Drug Application (ANDA) from a generic competitor. If the generic filer challenges a patent, they can submit their application after 4 years, but the 5-year block remains.
- 3-Year Clinical Investigation Exclusivity (CIE): This is for “me-too” drugs or new versions of old drugs. If you conduct new clinical trials (not just bioavailability studies) that are essential to approval—like a new dosage form or a new indication—you get 3 years of protection.
This type of exclusivity is crucial for “professional services” within the pharma world. Just as we help clients level up their leads for professional services client acquisition, pharmaceutical companies use CIE to protect new delivery methods (like moving from a pill to a long-acting injection) to keep their market share from eroding.
One Per Market Exclusivity for Biologics and Biosimilars
Biologics are the “big leagues” of the pharma world. Because they are complex mixtures derived from living organisms, they are much harder to copy than simple chemical pills. Consequently, they get much longer protection under the Biologics Price Competition and Innovation Act (BPCIA).
- 12-Year Protection: A reference biologic product gets a total of 12 years of market exclusivity.
- 4-Year Data Exclusivity: For the first 4 years after approval, a biosimilar applicant cannot even submit their application to the FDA.
- The 351(k) Pathway: This is the “generic” version of the biologic approval process. Because of the 12-year window, biosimilar entry is often delayed significantly, allowing the innovator to dominate the market for over a decade.
For a deeper dive into these rules, check out the FDA Guidance on Biologic Exclusivity.
Strategic Drivers for Maximizing One Per Market Exclusivity
Winning the launch game isn’t just about getting approved; it’s about “stacking” your protections. Smart developers don’t just settle for 5 years; they look for ways to add months or years to that timeline.
According to Research on Market Exclusivity Determinants, first-in-class drugs often manage to secure nearly 15 years of monopoly through strategic planning.
Leveraging Orphan and Pediatric Extensions
Two of the most powerful tools in your arsenal are Orphan and Pediatric designations.
- 7-Year Orphan Drug Exclusivity: If your drug treats a rare disease (affecting fewer than 200,000 people in the US), you get 7 years of protection for that specific use. This is powerful because it can be “broken” only if a competitor proves their drug is clinically superior.
- 6-Month Pediatric Extension: This is the “cherry on top.” If the FDA issues a written request for you to study your drug in children and you provide acceptable data, they add 6 months to all existing patents and exclusivities for that drug.
Think about that: 6 months of a blockbuster drug’s revenue can be worth hundreds of millions of dollars. You can read the specific Pediatric Exclusivity Provisions to see how to qualify.
Incentives for Antibiotics and Tropical Diseases
Because the world desperately needs new ways to fight “superbugs,” the GAIN Act provides a massive incentive: the QIDP designation.
- 5-Year Extension: If your drug is a “Qualifying Infectious Disease Product,” you get an automatic 5-year extension added to your NCE or Orphan exclusivity.
- Priority Review Vouchers (PRV): For certain tropical diseases or rare pediatric conditions, you might even get a voucher that fast-tracks the FDA review of a different future drug. These vouchers are so valuable they are often sold between companies for over $100 million.
For more info, see the QIDP Designation Questions and Answers.
Global Protection: The EU ‘8+2+1’ Rule vs. US Standards
If you’re launching globally, you need to know that Europe plays by a different set of rules. While the US has a “patchwork” of different exclusivities, the EU uses a more streamlined formula known as the ‘8+2+1’ rule.
Data Exclusivity vs. Market Protection
In the EU, protection is split into two phases:
- 8 Years of Data Exclusivity: During this time, a generic company cannot even reference your clinical data to file their application.
- 2 Years of Market Protection: After the 8 years are up, a generic can file and get approved, but they cannot actually launch the product on the market.
- +1 Year Extension: If you find a significant new therapeutic indication during the first 8 years, you can get an extra year of market protection, bringing the total to 11 years.
This framework is governed by EU Orphan Medicine Rules and provides a very predictable timeline for developers.
Comparing Orphan Drug Exclusivity Durations
Orphan drugs are treated even more generously in Europe than in the US.
- EU: 10 years of market exclusivity (can be reduced to 6 years if the drug is “too profitable,” though this is rare).
- Pediatric Extension: If you complete a Pediatric Investigation Plan (PIP), that 10-year orphan period can be extended to 12 years.
This is a key difference. While the US gives 7 years, the EU gives 10-12. If you’re developing a rare disease treatment, your European strategy might actually be more lucrative than your US one in the long run. Check the UK Guidance on Data Exclusivity for post-Brexit specifics.
The Impact of Exclusivity on Market Entry and Pricing
Why does all this legal jargon matter? Because it dictates the price of your healthcare and the profitability of your business.
During the period of one per market exclusivity, a company has a legal monopoly. They can set prices based on the value the drug provides, rather than competing on cost. For example, when the Hepatitis C cure Sovaldi launched, it was priced at $84,000 for a 12-week course. Because it was protected by exclusivity, there were no cheaper alternatives, even though the actual manufacturing cost was less than $150.
Generic Entry and the 180-Day Reward
Once the “triple fence” of patents and exclusivity falls, the market changes overnight. Prices typically drop by 70-80% within the first year of generic entry.
To encourage generic companies to challenge weak patents, the US offers a “180-day exclusivity” to the first generic filer. This gives that one generic company a 6-month head start to be the only low-cost alternative on the market, which is incredibly profitable.
In our world of digital marketing, we see similar dynamics. If you don’t qualify B2B traffic with the right keywords, you might end up spending your budget competing with the “generics” of the ad world instead of owning your high-value niche.
Policy Debates and Future Reforms
There is a growing debate about whether exclusivity periods are too long. Some critics argue that “evergreening”—the practice of stacking minor patents and exclusivities to block competition for decades—is driving up healthcare costs.
Proposed reforms include Research on variable market exclusivity periods, where the length of your monopoly would be tied to how much you spent on R&D. If you make your money back quickly, your exclusivity ends sooner. If you’re working on a neglected disease with low profits, your exclusivity could be extended.
Frequently Asked Questions about Market Protection
What is the difference between data exclusivity and market protection?
Data exclusivity (usually 8 years in the EU or 4 years for biologics in the US) prevents a competitor from using your lab and trial results to prove their own product works. Market protection (the extra 2 years in the EU) allows them to use your data to get their drug ready, but they are forbidden from actually selling it until the clock runs out.
How does pediatric exclusivity attach to existing patents?
Pediatric exclusivity is unique because it doesn’t stand on its own. It’s a “booster.” It adds 6 months to the end of every patent and every other type of regulatory exclusivity listed in the Orange Book for that specific drug. If you have three patents and one NCE exclusivity, all four get a 6-month extension.
Can a drug have multiple orphan exclusivities?
Yes! If a drug is approved for two different rare diseases, it can have two separate 7-year (US) or 10-year (EU) periods of orphan exclusivity. These periods usually overlap, but they protect each specific indication separately.
Conclusion
At the end of the day, one per market exclusivity is about one thing: balance. It’s the reward for the “rocket science” of drug development—the high-risk, high-cost journey of bringing a new molecule to life. Without these protections, the “2 in 10” drugs that actually turn a profit wouldn’t be enough to sustain the industry.
At Yael Consulting, we take this principle of exclusivity and apply it to your business growth. We believe that if we are helping you dominate your market through Google Ads, we shouldn’t be helping your competitor do the same thing. That’s why we offer our own version of one per market exclusivity—we only work with one client per industry per market.
Whether you are mastering lead generation in financial services or launching a new e-commerce brand, you deserve a partner who is as committed to your monopoly as you are.
Ready to see how we can help you own your market? Request a Free Google Ads Analysis today. We’ll give you a 15-minute, no-BS look at your account and show you exactly where you can build your own “exclusivity” in the digital space.


Ex Special-Ops commander turned Google Ads expert and online marketing consultant. In minutes I will show you exactly how I will improve your profits (no fluff), backed by a 30-day guarantee. Feel free to contact me.
Lior is an expert in online marketing, strategy, operations, and technology. In his experience with diverse industries, military, and small and fortune-500 companies, he personally increased sales and productivity, built reporting platforms, and cut wasteful costs, all to ultimately hit company goals.
Lior has passion for learning, curiosity, and genuine commitment to get results. He enjoys working with high-performance and results-driven teams and performs best in environments that strive for excellence.
Specialties: Search Engine Marketing (SEM, PPC, Paid Search), Google Adwords, Bing-Yahoo Marketing, Landing Page Optimization. Data, ROI, and LTV Analytics, Report and Process Automation.